Children’s Robitussin products recalled over incorrect dosing cups
LOS ANGELES - GSK Consumer Healthcare is voluntarily recalling two lots of Children’s Robitussin.
A Children's Robitussin Honey Cough and Chest Congestion DM and one lot of Children's Dimetapp Cold and Cough were recalled due to the inclusion of incorrect dosing cups.
The recall is limited to the lots listed below:
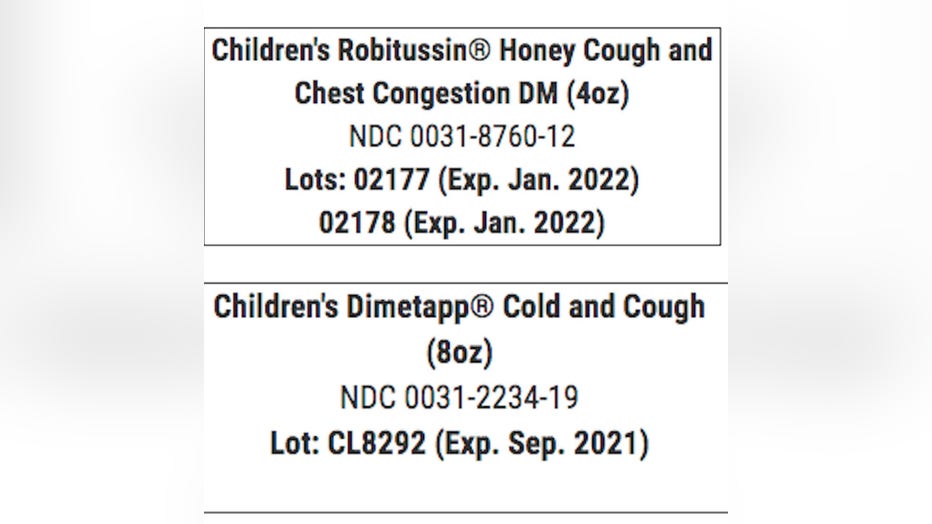
The safety alert was released by the Food and Drug Administration (FDA) on June 19.
During the review of the packaging documents for the products, GSK discovered that the dosing cups for the Children's Robitussin® Honey product were missing the 5 mL and 10 mL graduations, while the dosing cups for the Children's Dimetapp® product were missing the 10 mL graduation. The dosing cups packaged with both products only had the 20 mL graduation, the memo read.
GSK said they made the decision due to the “inclusion of incorrect dosing cups, posing a potential risk of accidental overdose.” In the statement to Fox TV, they added, “The safety of our consumers and product quality are our utmost priorities at GSK.”
RELATED: Contigo water bottles sold at Walmart, Target again recalled over choking hazard
The lots were distributed nationwide between Feb. 5, 2020 and June 3, 2020 within the United States.
According to the recall announcement, GSK Consumer Healthcare has notified wholesalers, distributors and retailers.
GSK Consumer Healthcare has not received any reports of adverse events related to the recalled products or consumer complaints regarding the incorrect dosing cups supplied with the products as of the recall announcement, the statement read.
RELATED: Nearly 43,000 pounds of ground beef recalled due to E. coli concerns
The FDA announced that there is a “potential risk of accidental overdose if caregivers dispensing the syrup do not notice the discrepancies between the graduations printed on the dosing cups and the indicated amounts to be administered.”

Robitussin cough and cold products labels and dosing cups
Consumers should contact their physician or health care provider if they have experienced any problems that may be related to taking or using the products.


