COVID-19 vaccine should be ready by the end of the year, health officials say

The recommendation for travelers to do their part to protect others from COVID-19
Justin Matthews reports
TAMPA, Fla. - The COVID-19 vaccine is waiting for its emergency use authorization by the Food and Drug Administration, and health officials anticipate that happening within the next few weeks.
"We’re looking at having multiple vaccines FDA approved and close to 40 million doses of FDA approved vaccine releasable by the end of this year," said Alex Azar, the U.S. Health and Human Services Secretary.
Despite the vaccine being on the horizon, Azar says the public needs to remain vigilant.
"Be washing their hands, watching their distance, wearing their face coverings when they can’t watch their distance because those bright days are ahead and we want everyone to be there to get their vaccination,” Azar said.
Featured
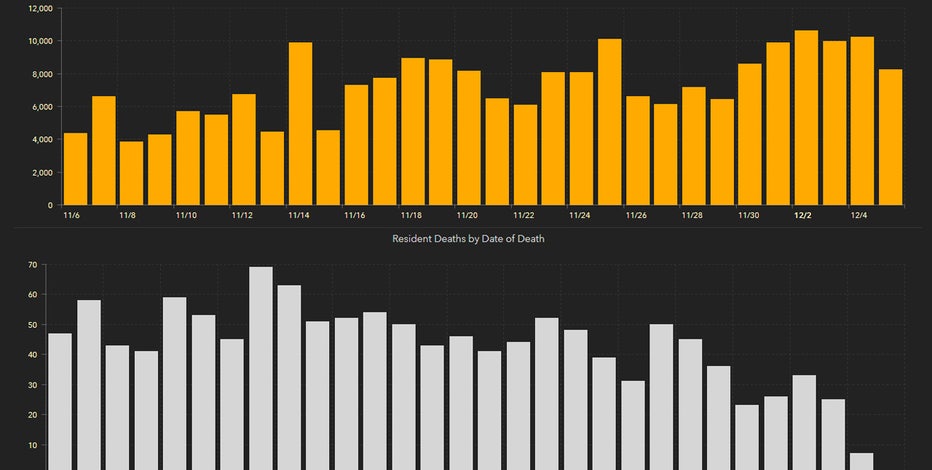
8,436 new Florida coronavirus cases reported Sunday; 93 new deaths
The Florida Department of Health says the number of known cases of COVID-19 in the state rose by 8,436 Sunday. According to the state's daily update, the total number of cases in Florida is now 1,058,074.
Also, the Centers for Disease Control and Prevention released a new set of guidelines for people traveling for the holidays. To help stop the spread, the CDC recommends travelers get tested for COVID-19 one to two days before their trip, and three to five days after their trip.